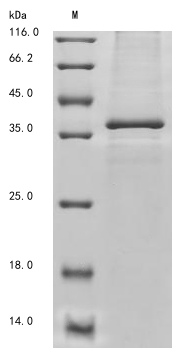
The recombinant human DNASE1L3 protein synthesis in baculovirus cells necessitates the incorporation of a DNA fragment encoding the human DNASE1L3 protein (21-305aa) into a plasmid vector along with the N-terminal 10xHis and C-terminal Myc-tag gene, followed by the transformation of this vector into baculovirus cells. After screening for positive cells, they are cultured and induced to express the DNASE1L3 protein. Cell lysis is performed to gather the recombinant human DNASE1L3 protein, which undergoes affinity purification and is then analyzed using SDS-PAGE and subsequent staining of the gel with Coomassie Brilliant Blue. The purity of the resulting recombinant human DNASE1L3 protein reaches up to 85%.
DNASE1L3, a member of the DNase family, functions as an endonuclease that degrades DNA during apoptosis [1]. DNASE1L3 has been suggested to interact with DFFB in DNA fragmentation during apoptosis [2]. DNASE1L3 was found to have both nuclear and extranuclear localization, indicating its diverse functions [3]. DNASE1L3 can degrade DNA complexed with proteins, like histone, compared with DNase1 in vitro [4]. The nuclease activity of DNASE1L3 is required to release DNA-binding proteins involved in IL-1β processing, indicating DNASE1L3's role in regulating inflammasome-dependent cytokine secretion [5].
DNASE1L3 has also been associated with immune response-related neutrophil activation and the regulation of acute inflammatory response and neutrophil-mediated cytotoxicity [6]. DNASE1L3 proteins contain an additional C-terminal domain of 21 amino acids rich in basic amino acids lysine and arginine [7]. DNASE1L3 may relieve cytoplasmic DNA accumulation under DNA damage response (DDR) activation [8]. The serum concentration of DNASE1L3 protein is reported to decrease in autoimmune diseases, suggesting DNASE1L3's involvement in their development [9]. DNASE1L3 inhibits hepatocellular carcinoma proliferation, invasion, and metastasis by interacting with β‐catenin to promote its ubiquitin degradation pathway [10].
References:
[1] J. Zochling, F. Newell, J. Charlesworth, P. Leo, J. Stankovich, A. Cortéset al., An immunochip-based interrogation of scleroderma susceptibility variants identifies a novel association at dnase1l3, Arthritis Research & Therapy, vol. 16, no. 5, 2014. https://doi.org/10.1186/s13075-014-0438-8
[2] D. Han, M. Ni, R. Chan, V. Chan, K. Lui, R. Chiuet al., The biology of cell-free dna fragmentation and the roles of dnase1, dnase1l3, and dffb, The American Journal of Human Genetics, vol. 106, no. 2, p. 202-214, 2020. https://doi.org/10.1016/j.ajhg.2020.01.008
[3] B. Lazzaretto and B. Fadeel, Intra- and extracellular degradation of neutrophil extracellular traps by macrophages and dendritic cells, The Journal of Immunology, vol. 203, no. 8, p. 2276-2290, 2019. https://doi.org/10.4049/jimmunol.1800159
[4] S. Inokuchi, H. Mitoma, S. Kawano, S. Nakano, M. Ayano, Y. Kimotoet al., Homeostatic milieu induces production of deoxyribonuclease 1–like 3 from myeloid cells, The Journal of Immunology, vol. 204, no. 8, p. 2088-2097, 2020. https://doi.org/10.4049/jimmunol.1901304
[5] G. Shi, K. Abbott, W. Wu, R. Salter, & P. Keyel, Dnase1l3 regulates inflammasome-dependent cytokine secretion, Frontiers in Immunology, vol. 8, 2017. https://doi.org/10.3389/fimmu.2017.00522
[6] Z. Deng, M. Xiao, D. Du, N. Luo, D. Liu, T. Liuet al., Dnase1l3 as a prognostic biomarker associated with immune cell infiltration in cancer, Oncotargets and Therapy, vol. Volume 14, p. 2003-2017, 2021. https://doi.org/10.2147/ott.s294332
[7] M. Napirei, S. Wulf, D. Eulitz, H. Mannherz, & T. Kloeckl, Comparative characterization of rat deoxyribonuclease 1 (dnase1) and murine deoxyribonuclease 1-like 3 (dnase1l3), Biochemical Journal, vol. 389, no. 2, p. 355-364, 2005. https://doi.org/10.1042/bj20042124
[8] D. Guo, D. Ma, P. Liu, J. Lan, Z. Liu, & Q. Liu, Dnase1l3 arrests tumor angiogenesis by impairing the senescence-associated secretory phenotype in response to stress, Aging, vol. 13, no. 7, p. 9874-9899, 2021. https://doi.org/10.18632/aging.202740
[9] S. Wang, H. Ma, X. Li, X. Mo, H. Zhang, Y. Denget al., Dnase1l3 as an indicator of favorable survival in hepatocellular carcinoma patients following resection, Aging, vol. 12, no. 2, p. 1171-1185, 2020. https://doi.org/10.18632/aging.102675
[10] B. Li, Y. Ge, W. Yan, B. Gong, K. Cao, R. Zhaoet al., dnase1l3 inhibits proliferation, invasion and metastasis of hepatocellular carcinoma by interacting with β‐catenin to promote its ubiquitin degradation pathway, Cell Proliferation, vol. 55, no. 9, 2022. https://doi.org/10.1111/cpr.13273